Water
Studying water in nano/micro-scale
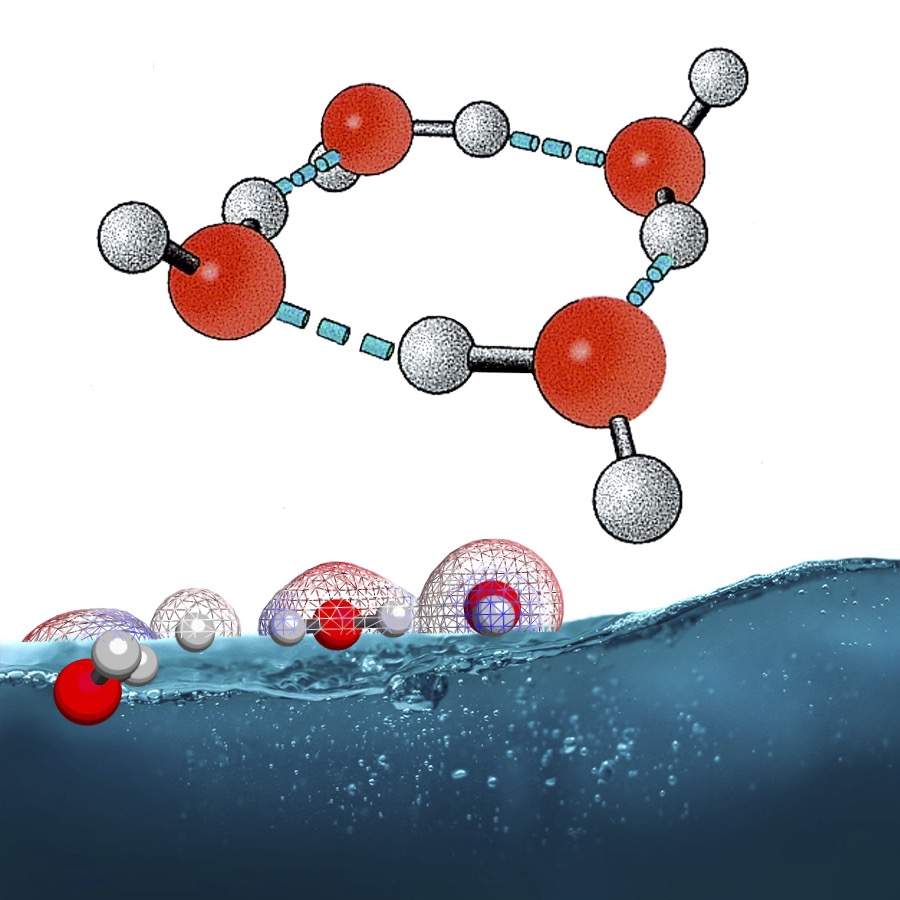
Water, with its seemingly simple molecular structure, holds a remarkable complexity at the molecular level due to its ability to form intricate hydrogen-bonding networks. Especially for water confined in nano/ micro-scale regions, we need to go beyond continuum models based on electrostatics or fluid-dynamics. The accuracy of simulations for water is of great importance in understanding problems like solvation dynamics and protein folding.
Problems of interest
- Modeling water in biological environments
- Proton transfer/ acid-base chemistry in water
- Reactions by solvated electrons in water
- Ice/ clathrate structures as graphs
- Improved continuum models at micro/ nanoscale incorporating quantum mechanical effects